Articles
- Page Path
- HOME > Res Vestib Sci > Volume 20(2); 2021 > Article
-
Original Article
재발성 양성돌발체위현기증의 임상적 특성: 후향적 코호트 연구 -
김재명1,*, 조방훈2,*, 이종경3, 김명규1, 이승한1

- Clinical Characteristics of Recurrent Benign Paroxysmal Positional Vertigo: A Retrospective Cohort Study
-
Jae-Myung Kim1,*, Bang-Hoon Cho2,*, Jong-Kyung Lee3, Myeong-Kyu Kim1, Seung-Han Lee1

-
Research in Vestibular Science 2021;20(2):45-50.
DOI: https://doi.org/10.21790/rvs.2021.20.2.45
Published online: June 14, 2021
1Department of Neurology, Chonnam National University Medical School and Chonnam National University Hospital, Gwangju, Korea
2Department of Neurology, Korea University College of Medicine, Korea University Anam Hospital, Seoul, Korea
3Department of Neurology, Happy View Hospital, Gwangju, Korea
- *Corresponding Author: Seung-Han Lee Department of Neurology, Chonnam National University Hospital, 42 Jebong-ro, Dong-gu, Gwangju 61469, Korea Tel: +82-62-220-6274 Fax: +82-62-228-3461 E-mail: nrshlee@chonnam.ac.kr
- *These authors contributed equally to this study.
• Received: May 14, 2021 • Revised: May 22, 2021 • Accepted: May 24, 2021
Copyright © 2021 by The Korean Balance Society. All rights reserved.
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0) which permits unrestricted non-Commercial use, distribution, and re-production in any medium, provided the original work is properly cited.
- 3,909 Views
- 68 Download
Abstract
-
Objectives
- Benign paroxysmal positional vertigo (BPPV) is a potentially recurrent disease even after successful canalith repositioning maneuvers. However, the exact recurrence rate or any clinical factors affecting the recurrence of BPPV still need to be elucidated.
-
Methods
- We recruited patients diagnosed as BPPV in a tertiary hospital for 3 years. We retrospectively reviewed the clinical information of the patients including baseline demographics, comorbidities and predisposing factors through the electronic medical records. We performed a telephone survey or direct interview 3 to 6 years later from the initial diagnosis of BPPV was made. To determine the factors associated with the recurrence, we divided study population into two subgroups; ‘recurrence group’ vs. ‘recurrence-free group.’ Then, intergroup comparative analyses were performed.
-
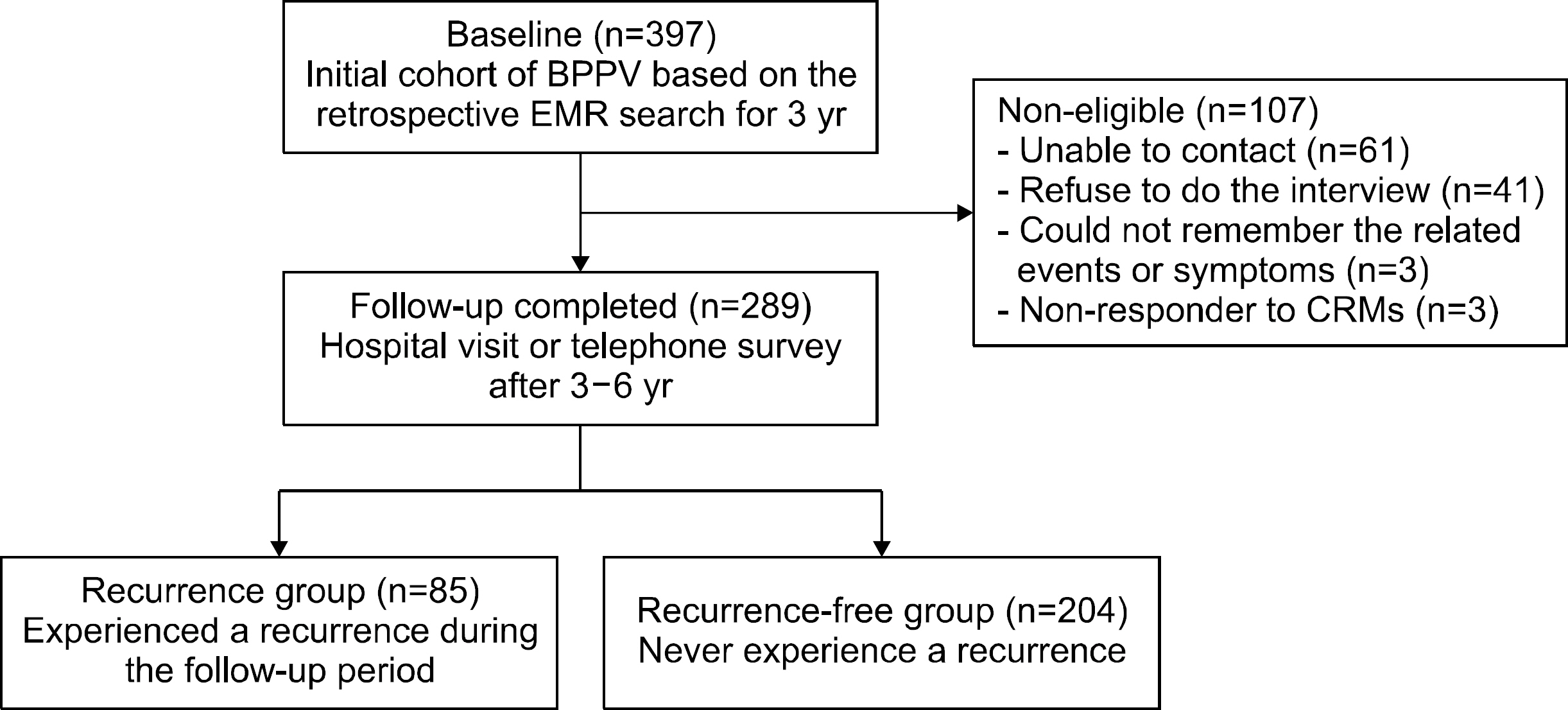
Results
- Among 397 patients who were originally eligible for the study, we performed a telephone survey or direct interview in 289 patients (72.8%) to determine the recurrence of BPPV. The overall recurrence rate was 29.4% (85 of 289). Baseline demographics except female gender (p=0.014) were not different between subgroups. Neither clinical characteristics nor vascular comorbidities were associated with the recurrence. However, patients with low bone mineral density (BMD; T-score below ‒1, osteopenia/osteoporosis) showed significantly higher recurrence than those with normal BMD (40.6% vs. 0%, p=0.009).
-
Conclusions
- Female gender and low BMD (T-score below ‒1) were associated with the recurrence of BPPV in this study. Further researches in various clinical settings with larger sample size are warranted to identify the factors affecting the relapse of BPPV.
INTRODUCTION
MATERIALS AND METHODS
Fig. 1.Study flow sheet of the study participants. BPPV, benign paroxysmal positional vertigo; EMR, electronic medical records; CRM, canalith repositioning maneuver.

RESULTS
Table 1.Demographic characteristics of the recruited BPPV patients
| Characteristic | Recurrence group (n=85) | Recurrence-free group (n=204) | p-value |
|---|---|---|---|
| Age (yr) | 60.1±12.4 | 59.4±13.4 | 0.68 |
| Female sex | 61 (71.8) | 113 (55.4) | 0.014* |
| Hypertension | 30 (35.3) | 72 (35.3) | >0.999 |
| Diabetes mellitus | 11 (12.9) | 28 (13.7) | >0.999 |
| Previous stroke | 4 (4.7) | 6 (2.9) | 0.46 |
| Smoking | 4 (4.7) | 19 (9.3) | 0.19 |
| Recent head traumaa) | 3 (3.5) | 10 (4.9) | 0.61 |
| Recent URI | 4 (4.7) | 10 (4.9) | 0.94 |
| Inner ear symptomsb) | 11 (12.9) | 31 (15.2) | 0.62 |
Table 2.Comparative analysis of the recurrence rate of benign paroxysmal positional vertigo
| Variable | No. of patients | Recurrence | p-value |
|---|---|---|---|
| Semicircular canal involvement | 0.915 | ||
| Horizontal canal | 179 | ||
| Canalolithiasis | 78 | 25 (32.1) | |
| Cupulolithiasis | 101 | 29 (28.7) | |
| Posterior canal | 92 | 27 (29.3) | |
| Anterior canal | 4 | 1 (25.0) | |
| Multicanal | 14 | 3 (21.4) | |
| No. of canal | 0.764 | ||
| Single | 275 | 82 (29.8) | |
| Multiple | 14 | 3 (21.4) | |
| Bone mineral densitya) | 0.009* | ||
| Normal | 13 | 0 (0) | |
| Lowb) | 32 | 13 (40.6) |
DISCUSSION
SUPPLEMENTARY MATERIALS
Supplementary material.
- 1. Lee SH, Kim JS. Benign paroxysmal positional vertigo. J Clin Neurol 2010;6:51–63.ArticlePubMedPMC
- 2. Kim JS, Zee DS. Clinical practice. Benign paroxysmal positional vertigo. N Engl J Med 2014;370:1138–47.ArticlePubMed
- 3. Lopez-Escamez JA, Gamiz MJ, Fernandez-Perez A, Gomez-Fiñana M. Long-term outcome and health-related quality of life in benign paroxysmal positional vertigo. Eur Arch Otorhinolaryngol 2005;262:507–11.ArticlePubMed
- 4. Dieterich M, Staab JP. Functional dizziness: from phobic postural vertigo and chronic subjective dizziness to persistent postural-perceptual dizziness. Curr Opin Neurol 2017;30:107–13.ArticlePubMed
- 5. Yu S, Liu F, Cheng Z, Wang Q. Association between osteoporosis and benign paroxysmal positional vertigo: a systematic review. BMC Neurol 2014;14:110. ArticlePubMedPMC
- 6. Jeong SH, Kim JS. Impaired calcium metabolism in benign paroxysmal positional vertigo: a topical review. J Neurol Phys Ther 2019;43(Suppl 2):S37–41.ArticlePubMed
- 7. Neuhauser HK. The epidemiology of dizziness and vertigo. Handb Clin Neurol 2016;137:67–82.ArticlePubMed
- 8. Kim JY, Ko JS, Lee HJ, Hur DG, Ahn SK. Long-term follow-up of patients with benign paroxysmal positional vertigo. Res Vestib Sci 2015;14:83–6.
- 9. Rhim GI. Long-term outcomes of canalith repositioning for benign paroxysmal positional vertigo: Kaplan-Meier estimate. Res Vestib Sci 2016;15:17–21.
- 10. Li S, Wang Z, Liu Y, Cao J, Zheng H, Jing Y, et al. Risk factors for the recurrence of benign paroxysmal positional vertigo: a systematic review and meta-analysis. Ear Nose Throat J 2020 Aug 10;[Epub]. https://doi.org/10.1177/0145561320943362.Article
- 11. Chen J, Zhang S, Cui K, Liu C. Risk factors for benign paroxysmal positional vertigo recurrence: a systematic review and meta-analysis. J Neurol 2020 Aug 24;[Epub]. https://doi.org/10.1007/s00415-020-10175-0.Article
- 12. Luryi AL, Lawrence J, Bojrab DI, LaRouere M, Babu S, Zappia J, et al. Recurrence in benign paroxysmal positional vertigo: a large, single-institution study. Otol Neurotol 2018;39:622–7.ArticlePubMed
- 13. Lee SH, Choi KD, Jeong SH, Oh YM, Koo JW, Kim JS. Nystagmus during neck flexion in the pitch plane in benign paroxysmal positional vertigo involving the horizontal canal. J Neurol Sci 2007;256:75–80.ArticlePubMed
- 14. Epley JM. The canalith repositioning procedure: for treatment of benign paroxysmal positional vertigo. Otolaryngol Head Neck Surg 1992;107:399–404.ArticlePubMed
- 15. Nunez RA, Cass SP, Furman JM. Short- and long-term outcomes of canalith repositioning for benign paroxysmal positional vertigo. Otolaryngol Head Neck Surg 2000;122:647–52.ArticlePubMed
- 16. Parnes LS, Agrawal SK, Atlas J. Diagnosis and management of benign paroxysmal positional vertigo (BPPV). CMAJ 2003;169:681–93.PubMedPMC
- 17. Zhu CT, Zhao XQ, Ju Y, Wang Y, Chen MM, Cui Y. Clinical characteristics and risk factors for the recurrence of benign paroxysmal positional vertigo. Front Neurol 2019;10:1190. ArticlePubMedPMC
- 18. Kao CL, Cheng YY, Leu HB, Chen TJ, Ma HI, Chen JW, et al. Increased risk of ischemic stroke in patients with benign paroxysmal positional vertigo: a 9-year follow-up nationwide population study in Taiwan. Front Aging Neurosci 2014;6:108. ArticlePubMedPMC
- 19. Prokopakis EP, Chimona T, Tsagournisakis M, Christodoulou P, Hirsch BE, Lachanas VA, et al. Benign paroxysmal positional vertigo: 10-year experience in treating 592 patients with canalith repositioning procedure. Laryngoscope 2005;115:1667–71.ArticlePubMed
- 20. Gordon CR, Levite R, Joffe V, Gadoth N. Is posttraumatic benign paroxysmal positional vertigo different from the idiopathic form? Arch Neurol 2004;61:1590–93.ArticlePubMed
- 21. Suarez H, Alonso R, Arocena M, Suarez A, Geisinger D. Clinical characteristics of positional vertigo after mild head trauma. Acta Otolaryngol 2011;131:377–81.ArticlePubMed
- 22. Ahn SK, Jeon SY, Kim JP, Park JJ, Hur DG, Kim DW, et al. Clinical characteristics and treatment of benign paroxysmal positional vertigo after traumatic brain injury. J Trauma 2011;70:442–6.ArticlePubMed
- 23. Jeong SH, Choi SH, Kim JY, Koo JW, Kim HJ, Kim JS. Osteopenia and osteoporosis in idiopathic benign positional vertigo. Neurology 2009;72:1069–76.ArticlePubMed
- 24. Johnsson LG, Rouse RC, Wright CG, Henry PJ, Hawkins JE Jr. Pathology of neuroepithelial suprastructures of the human inner ear. Am J Otolaryngol 1982;3:77–90.ArticlePubMed
- 25. Walther LE, Wenzel A, Buder J, Bloching MB, Kniep R, Blödow A. Detection of human utricular otoconia degeneration in vital specimen and implications for benign paroxysmal positional vertigo. Eur Arch Otorhinolaryngol 2014;271:3133–8.ArticlePubMed
- 26. Yamanaka T, Shirota S, Sawai Y, Murai T, Fujita N, Hosoi H. Osteoporosis as a risk factor for the recurrence of benign paroxysmal positional vertigo. Laryngoscope 2013;123:2813–6.ArticlePubMed
- 27. Vibert D, Sans A, Kompis M, Travo C, Muhlbauer RC, Tschudi I, et al. Ultrastructural changes in otoconia of osteoporotic rats. Audiol Neurootol 2008;13:293–301.ArticlePubMed
- 28. Kim HJ, Song JM, Zhong L, Yang X, Kim JS. Questionnaire-based diagnosis of benign paroxysmal positional vertigo. Neurology 2020;94:e942–9.ArticlePubMed
- 29. Lapenna R, Faralli M, Del Zompo MR, Cipriani L, Mobaraki PD, Ricci G. Reliability of an anamnestic questionnaire for the diagnosis of benign paroxysmal positional vertigo in the elderly. Aging Clin Exp Res 2016;28:881–8.ArticlePubMed
- 30. von Brevern M, Radtke A, Lezius F, et al. Epidemiology of benign paroxysmal positional vertigo: a population based study. J Neurol Neurosurg Psychiatry 2007;78:710–5.ArticlePubMed
REFERENCES
Figure & Data
References
Citations
Citations to this article as recorded by 

- Figure
- We recommend
- Related articles
-
- Canal Conversion and Reentry of Otolith in Benign Paroxysmal Positional Vertigo
- Clinical Characteristics of Benign Paroxysmal Positional Vertigo Positive on Bilateral Dix-Hallpike Test
- Treatment in Benign Paroxysmal Positional Vertigo: Factors that Affect Successful Treatment Outcome
- The Head-Bending Test in Posterior Semicircular Canal Benign Paroxysmal Positional Vertigo
- Intravenous Zoledronic Acid in Elderly Patients with Benign Paroxysmal Positional Vertigo and Osteoporosis

 KBS
KBS
 PubReader
PubReader ePub Link
ePub Link Cite
Cite