Abstract
-
Objectives:
- Light cupula is characterized by persistent geotropic direction-changing positional nystagmus in a supine head-roll test. The purpose of this study is to investigate if hearing level is influenced by the change of head position in light cupula under the assumption that relative density difference similarly occurs between the tectorial membrane and endolymph.
-
Methods:
- Twelve patients with unilateral light cupula who underwent positional audiometry were included in this study. Pure tone thresholds were compared among three head positions.
-
Results:
- Hearing threshold in pure tone audiometry (PTA) of the affected ear was not different from that of the healthy ear. PTA thresholds of the affected side were not significantly different in three head positions; upright seating, cochlear apex-up, and cochlear apex-down positions.
-
Conclusions:
- Although positional change of nystagmus direction is the most significant clinical feature of light cupula, positional change of hearing level was not observed in those patients. The lack of positional influence on hearing may be explained as follows: (1) the heavier endolymph phenomenon occurs only in the vestibular end organ without involving the cochlea; (2) the light cupula phenomenon is more likely to occur due to light debris mechanism rather than heavier endolymph or lighter cupula mechanism; and (3) the effects of light cupula could be modified by outer hair cells, which work for tuning in the cochlea, even though light tectorial membrane or heavy endolymph occurs.
-
Keywords: Light cupula; Positional audiometry; Vertigo; Nystagmus; Hearing loss
-
중심단어: 가벼운팽대부마루, 체위성청력검사, 현훈, 안진, 난청
INTRODUCTION
Since Hiruma and Numata’s first report [1] in the English literature, studies on the light cupula phenomenon have been conducted by researchers [2-7]. In the light cupula, specific gravity of the cupula is lower than that of the surrounding endolymphatic fluid, which either activates or inhibits hair cells due to the deflection of the cupula caused by gravity-dependent buoyant force. The light cupula is characterized by a typical positional nystagmus, which demonstrates persistent geotropic direction-changing positional nystagmus (DCPN) in a supine head-roll test. Although pathophysiological mechanism of the light cupula phenomenon is in debate, three hypotheses have been proposed; (1) light debris hypothesis, (2) lighter cupula phenomenon, and (3) heavier endolymph phenomenon. If the density difference between the cupula and endolymph is caused by alteration in chemical composition of the cupula or endolymph, it is reasonable to assume that the tectorial membrane, known as the cochlear counterpart to the cupula, can also be influenced based on the head position in the gravitational plane. This is because endolymph circulates between the cochlea and vestibular organs within the endolymphatic membrane.
In this study, a positional audiometry was conducted in patients with light cupula and investigation carried out on if hearing level is influenced by a head position in those patients.
MATERIALS AND METHODS
The study used twelve patients who were diagnosed with unilateral light cupula between March 2018 and August 2018. The exclusion criteria for the study was patients with other accompanying inner ear disorders such as sudden sensorineural hearing loss, otitis media, vestibular neuritis, Menière’s disease, and Ramsay Hunt syndrome. Also, those who had recent head trauma, central nervous system disorders such as migraine and cerebellar infarct, or those who had undergone recent surgery, were excluded. All patients underwent a detailed medical history taking. Neurological examination revealed no abnormalities in any patient. Eye movement was examined in various head positions and the results were recorded using goggles with an infrared camera (InView, Micromedical, Chatham, IL, USA; Easy-eyes, SLMED, Seoul, Korea). The diagnostic criteria for light cupula were the presence of persistent geotropic DCPN, which lasted for more than 2 minutes in a supine head-roll test, and the presence of a null plane where nystagmus disappeared with slight head rotation either to the right or to left. Determination of the affected side was made based on identification of a null plane, i.e., the side of a null plane corresponds to the affected side [2,8,9].
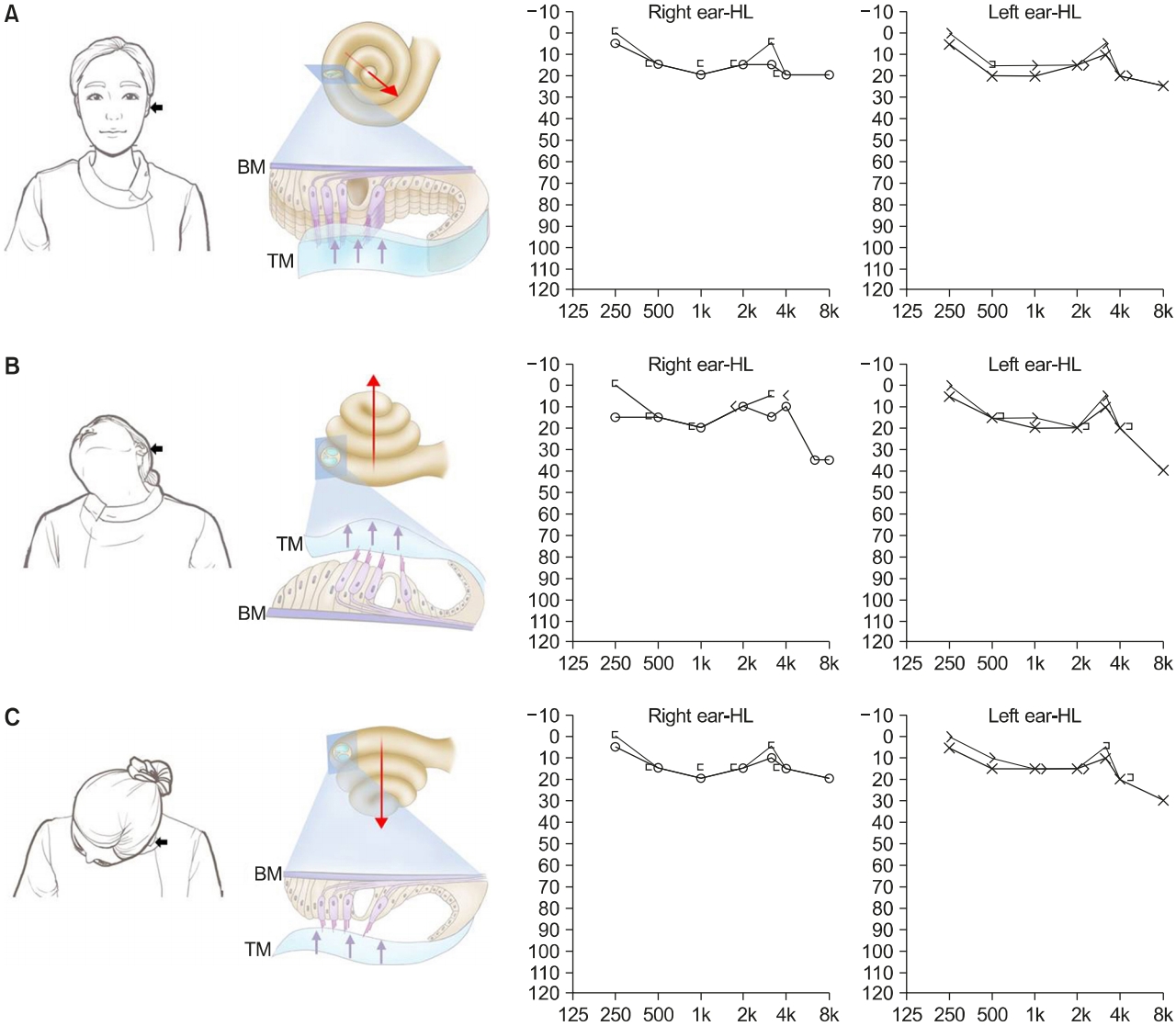
Positional audiometry was conducted in the patients with light cupula immediately after the diagnosis of light cupula. A pure tone audiometry (PTA) was performed in an upright seating position (Fig. 1A, left panel), measuring both air and bone conduction thresholds with appropriate masking in the usual way. In this position, cochlear apex of the affected ear directs to the anterolateral and inferiorly (Fig. 1A, middle panel) [10]. The patient’s head was turned 45° to the unaffected side and tilted backward (Fig. 1B, left panel). In this position, cochlear apex of the affected ear directs superiorly (Fig. 1B, middle panel), and PTA was performed. The patient’s head was thereafter tilted forward while turning 45° to the unaffected side (Fig. 1C, left panel). In this position, cochlear apex of the affected ear directs inferiorly (Fig. 1C, middle panel), and PTA was performed. We assumed that PTA thresholds, compared to that in upright seating position, would be higher in cochlear apex-up or cochlear apex-down position because the subtectorial space would be wider or narrower than that in upright seating position.
This study was approved by the Institutional Review Board of Konkuk University Medical Center (No. KUH1110078) and written informed consents were obtained from all participants.
RESULTS
Clinical profiles and results of positional nystagmus were shown in Table 1. The male-to-female ratio was 2:10 (age range, 43 to 77 years). A null plane, at which nystagmus disappears, was clearly identified in all 12 patients (right in four patients and left in eight patients), and the affected side was determined as the same side with that of a null plane. To examine bowing and leaning nystagmus, the patients were subjected to bowing (90°) and leaning (45°) in a sitting position. The direction of bowing nystagmus corresponded to the affected side while that of leaning nystagmus was opposite to the affected side, which was consistent with previous observations [2,8,9,11].
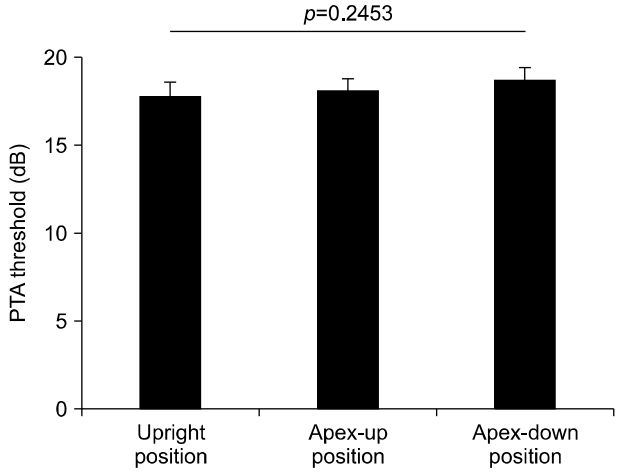
Under the assumption that specific gravity of the tectorial membrane, a cochlear counterpart to the cupula, is lower than that of surrounding endolymph in patients with the light cupula, positional audiometry was performed at three positions described in Materials and Methods section; (1) upright position (Fig. 1A), (2) cochlear apex-up position (Fig. 1B), and (3) cochlear apex-down position (Fig. 1C). In an upright seating position, the cochlear apex directs anterolaterally and slightly inferiorly (Fig. 1A, middle panel, long arrow). With respect to the direction of gravity, the tectorial membrane may deflect to the direction along the cochlear duct (Fig. 1A, middle panel, short arrows). The right panel of Fig. 1A depicts an example PTA at this position in patient 1, who had light cupula on the left side, from Table 1. The cochlear apex-up (Fig. 1B, middle panel, long arrow) position was made when the patient’s head turned 45° to the unaffected side and tilted 90° backward (Fig. 1B, left panel), and the tectorial membrane may deflect away from the stereocilia of the hair cells (Fig. 1B, middle panel, short arrows). An example PTA at this position is shown (Fig. 1B, right panel). The cochlear apex-down (Fig. 1C, middle panel, long arrow) position was made when the patient’s head turned 45° to the unaffected side and tilted 90° forward (Fig. 1C, left panel), and the tectorial membrane may deflect to the stereocilia of the hair cells (Fig. 1C, middle panel, short arrows). An example PTA at this position is shown (Fig. 1C, right panel). Average of PTA threshold (at 500, 1,000, 2,000, and 4,000 Hz) at the upright seating position was 17.8 dB on the affected side and 18.3 dB on the unaffected side, and the difference was insignificant (p=0.913, Student t-test). PTA threshold at the upright position, cochlear apex-up position, and cochlear apex-down position was 17.81 dB, 18.13 dB, and 18.75 dB, respectively, and the difference was insignificant (p=0.2453, Friedman test; Fig. 2). The difference in PTA threshold of the affected side between cochlear apex-up and apexdown position was 10 dB or less at each frequency in all patients as shown in Table 2.
DISCUSSION
The hypotheses explaining light cupula phenomenon include “light debris hypothesis,” “heavier endolymph hypothesis,” and “lighter cupula hypothesis” [12]. Light debris hypothesis suggests that light debris are attached on the surface of lateral semicircular canal cupula, resulting in cupula lighter than the surrounding endolymph. Heavier endolymph hypothesis assumes that the density or specific gravity of the endolymph is higher due to alteration in chemical composition of the endolymph. Lighter cupula hypothesis assumes that density of the cupula is decreased by altered homeostasis of macromolecules forming the cupula, or an irreversible morphological change of the cupula (enlarged cupula). The density of the cupula is equal to that of the endolymph in a normal condition, and the activation of semicircular canals are gravity-independent. In a condition of light cupula, the characteristic positional nystagmus is attributed to the buoyant force inflicted on the cupula which is immersed within the endolymphatic space.
Endolymphatic fluids are circulating between the cochlea and the vestibular organs within the endolymphatic membranes. Thus, if “heavier endolymph hypothesis” is the underlying mechanism, it is reasonable to assume that light cupula phenomenon may also occur in the cochlea. It was established that extracellular matrices in the inner ear, including cupula in the semicircular canals, otolithic membrane in the utricle and saccule, and tectorial membrane in the cochlea, all share similar roles in sensing acceleration or vibration. In addition, these matrices form intimate contact with the stereocilia of the sensory hair cells in common, and generally serve to transmit primary stimulus to stereocilia. On the other hand, Goodyear and Richardson [13] reported that a progressive increase in compositional and structural complexity is observed, with the cupula being the simplest, followed by the otolithic membrane exhibiting an intermediate, and the tectorial membrane being the most elaborate of the three matrices. The tectorial membrane is composed of several types of collagen (type II, IX, and XI) and three noncollagenous glycoproteins; α-tectorin, β-tectorin, and otogelin [14-17]. The tectorial membrane is attached along its medial edge to the spiral limbus, stretches across the internal spiral sulcus, and lies over the sensory epithelium, attaching along its lower lateral surface to the tips of the hair bundles of the outer hair cells. It has been suggested that proper spacing between the tectorial membrane and the reticular lamina is essential for optimal mechanotransduction in hearing [14], from the observations that auditory thresholds are elevated in mice with an enlarged subtectorial space [18]. Moreover, the magnitude of fluid flow and thus stereocilia deflection are believed to strongly depend on the distance between the tectorial membrane and the reticular lamina [19], and the enlargement of subtectorial space may affect the temporal pattern of inner hair cell depolarization during one cycle [20]. So far, there are no reported studies on density difference between the tectorial membrane and endolymph, and the influence of density difference between them has not received much attention in most studies about micromechanics of the tectorial membrane. However, in a condition of light cupula caused by “heavier endolymph” or “lighter cupula” mechanism, it is possible that the subtectorial space can be altered according to the head position.
The concept of positional audiometry was first introduced in the diagnosis of perilymphatic fistula where the presence of air within the cochlea caused an impedance mismatch, reducing sound transmission through the perilymph [21,22]. Positional audiometry for perilymphatic fistula is based on the assumption that horizontal positioning of the patient with the affected ear uppermost may displace air bubbles within the perilymphatic space to the middle ear cavity, thereby improving the ability to hear. This study demonstrated that in patients with light cupula, hearing level was not worse in the affected ear compared to the unaffected ear, and PTA thresholds among three head positions (upright seating, cochlear apex-up, and cochlear apex-down positions) showed insignificant differences. It was assumed that if density of the tectorial membrane becomes relatively lower than that of surrounding endolymph in patients with light cupula, the subtectorial space would be widened at cochlear apex-up position (Fig. 1B) and narrowed at cochlear apex-down position (Fig. 1C), which may influence the results of PTA thresholds. However, positional audiometric test revealed that change in head position appeared to have little impact on hearing threshold. Although underlying mechanism is unknown, the lack of positional influence on hearing may be explained as follows: (1) the heavier endolymph phenomenon occurs only in the vestibular end organ without involving the cochlea; (2) the light cupula phenomenon is more likely to occur due to light debris mechanism rather than heavier endolymph or lighter cupula mechanism; and (3) the effects of light cupula could be modified by outer hair cells, which work for tuning in the cochlea, even though light tectorial membrane or heavy endolymph occurs.
In conclusion, positional change of PTA threshold was not observed in patients with light cupula even though positional change of nystagmus direction is the most significant clinical feature of light cupula phenomenon. The lack of positional influence on hearing can be explained as (1) the heavier endolymph phenomenon occurs only in the vestibular apparatus of the inner ear, or (2) the effect of light tectorial membrane or heavy endolymph phenomenon was reduced by the modification of outer hair cell function.
ARTICLE INFORMATION
-
No potential conflict of interest relevant to this article was reported.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (2019R1H1A1080123).
Fig. 1.Positional audiometry at different head positions in an example case (patient 1 in Table 1) of the left light cupula. In the middle panels, the tectorial membrane (TM) in the cochlea is depicted under the assumption that the density of TM is lower than that of surrounding endolymph. (A) At an upright seating position, the cochlear apex of the affected ear directs anterolaterally and slightly inferiorly (long arrow), and TM is deflected to longitudinal direction along the cochlear duct (short arrows). Hearing threshold of pure tone audiometry (PTA) at this position was 18.8 dB (average of 0.5, 1, 2, and 4 kHz) in the affected ear and 17.5 dB in the unaffected ear. (B) When the patient’s head was turned 45° to the unaffected side and tilted backward, cochlear apex of the affected ear directs superiorly (long arrow). TM is deflected away from the basilar membrane (BM; short arrows), widening the subtectorial space. PTA threshold at this position was 18.8 dB in the affected ear and 13.8 dB in the unaffected ear. (C) When the patient’s head was turned 45° to the unaffected side and tilted forward, cochlear apex of the affected ear directs inferiorly (long arrow). TM is deflected toward BM (short arrows), narrowing the subtectorial space. PTA threshold at this position was 16.3 dB in the affected ear and 16.3 dB in the unaffected ear. HL, hearing level.

Fig. 2.Pure tone audiometry (PTA) of the affected ear at different positions. PTA threshold was 17.81±0.81 dB at an upright seating position, 18.13±0.71 dB at the cochlear apex-up position, and 18.75±0.72 at the cochlear apex-down position, which was not significantly different (p=0.2453, Friedman test).
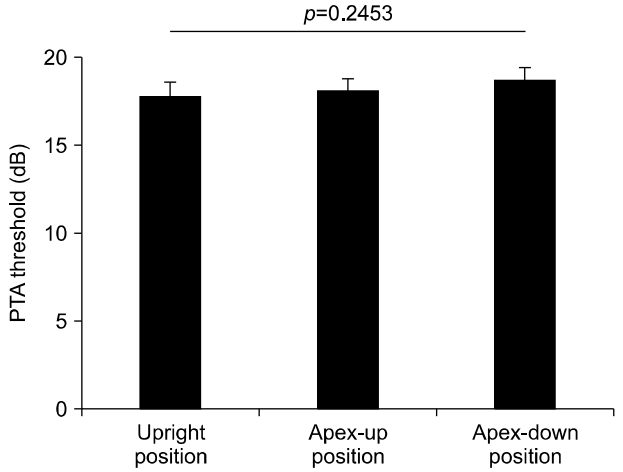
Table 1.Patients with light cupula showing persistent geotropic direction-changing positional nystagmus with a null plane
|
Patient No. |
Sex |
Age (yr) |
Bow |
Lean |
Side of null plane |
Affected side |
|
1 |
Female |
54 |
LB |
RB |
Left |
Left |
|
2 |
Female |
56 |
LB |
RB |
Left |
Left |
|
3 |
Female |
49 |
RB |
LB |
Right |
Right |
|
4 |
Male |
63 |
RB |
LB |
Right |
Right |
|
5 |
Female |
54 |
RB |
LB |
Right |
Right |
|
6 |
Female |
63 |
LB |
RB |
Left |
Left |
|
7 |
Female |
51 |
LB |
RB |
Left |
Left |
|
8 |
Female |
77 |
LB |
RB |
Left |
Left |
|
9 |
Male |
55 |
LB |
RB |
Left |
Left |
|
10 |
Female |
52 |
RB |
LB |
Right |
Right |
|
11 |
Female |
59 |
LB |
RB |
Left |
Left |
|
12 |
Female |
43 |
LB |
RB |
Left |
Left |
Table 2.Difference in pure tone audiometry between apex-up and apex-down positions at each frequency
|
Frequency (Hz) |
No. of patients (%)
|
|
≤10 dB |
>10 dB |
|
250 |
12 (100) |
0 (0) |
|
500 |
12 (100) |
0 (0) |
|
1,000 |
12 (100) |
0 (0) |
|
2,000 |
12 (100) |
0 (0) |
|
3,000 |
12 (100) |
0 (0) |
|
4,000 |
12 (100) |
0 (0) |
|
8,000 |
12 (100) |
0 (0) |
REFERENCES
- 1. Hiruma K, Numata T. Positional nystagmus showing neutral points. ORL J Otorhinolaryngol Relat Spec 2004;66:46–50.ArticlePubMed
- 2. Kim CH, Kim MB, Ban JH. Persistent geotropic direction-changing positional nystagmus with a null plane: the light cupula. Laryngoscope 2014;124:E15–9.ArticlePubMed
- 3. Bergenius J, Tomanovic T. Persistent geotropic nystagmus: a different kind of cupular pathology and its localizing signs. Acta Otolaryngol 2006;126:698–704.ArticlePubMed
- 4. Imai T, Matsuda K, Takeda N, Uno A, Kitahara T, Horii A, et al. Light cupula: the pathophysiological basis of persistent geotropic positional nystagmus. BMJ Open 2015;5:e006607. ArticlePubMedPMC
- 5. Ichijo H. Persistent direction-changing geotropic positional nystagmus. Eur Arch Otorhinolaryngol 2012;269:747–51.ArticlePubMedPDF
- 6. Schubert MC, Dunlap PM, Whitney SL. A case study of high-velocity, persistent geotropic nystagmus: is this BPPV? J Neurol Phys Ther 2017;41:182–6.ArticlePubMed
- 7. Shin JE, Jeong KH, Ahn SH, Kim CH. Conversion between geotropic and apogeotropic persistent direction-changing positional nystagmus. Acta Otolaryngol 2015;135:1238–44.ArticlePubMed
- 8. Kim CH, Shin JE, Kim YW. A new method for evaluating lateral semicircular canal cupulopathy. Laryngoscope 2015;125:1921–5.ArticlePubMed
- 9. Kim CH, Kim YG, Shin JE, Yang YS, Im D. Lateralization of horizontal semicircular canal canalolithiasis and cupulopathy using bow and lean test and head-roll test. Eur Arch Otorhinolaryngol 2016;273:3003–9.ArticlePubMedPDF
- 10. Schuknecht H. Pathology of the ear. 2nd ed. Malvern: Lea & Febiger; 1993.
- 11. Choi S, Choi HR, Nahm H, Han K, Shin JE, Kim CH. Utility of the bow and lean test in predicting subtype of benign paroxysmal positional vertigo. Laryngoscope 2018;128:2600–4.ArticlePubMed
- 12. Kim MB, Hong SM, Choi H, Choi S, Pham NC, Shin JE, et al. The light cupula: an emerging new concept for positional vertigo. J Audiol Otol 2018;22:1–5.ArticlePubMedPDF
- 13. Goodyear RJ, Richardson GP. Extracellular matrices associated with the apical surfaces of sensory epithelia in the inner ear: molecular and structural diversity. J Neurobiol 2002;53:212–27.ArticlePubMed
- 14. Gavara N, Manoussaki D, Chadwick RS. Auditory mechanics of the tectorial membrane and the cochlear spiral. Curr Opin Otolaryngol Head Neck Surg 2011;19:382–7.ArticlePubMedPMC
- 15. Goodyear RJ, Richardson GP. Structure, function, and development of the tectorial membrane: an extracellular matrix essential for hearing. Curr Top Dev Biol 2018;130:217–44.ArticlePubMed
- 16. Richardson GP, Lukashkin AN, Russell IJ. The tectorial membrane: one slice of a complex cochlear sandwich. Curr Opin Otolaryngol Head Neck Surg 2008;16:458–64.ArticlePubMedPMC
- 17. Sellon JB, Ghaffari R, Freeman DM. The tectorial membrane: mechanical properties and functions. Cold Spring Harb Perspect Med 2019;9:a033514. ArticlePubMedPMC
- 18. Legan PK, Lukashkina VA, Goodyear RJ, Lukashkin AN, Verhoeven K, Van Camp G, et al. A deafness mutation isolates a second role for the tectorial membrane in hearing. Nat Neurosci 2005;8:1035–42.ArticlePubMedPDF
- 19. Freeman DM, Weiss TF. Hydrodynamic forces on hair bundles at low frequencies. Hear Res 1990;48:17–30.ArticlePubMed
- 20. Smith ST, Chadwick RS. Simulation of the response of the inner hair cell stereocilia bundle to an acoustical stimulus. PLoS One 2011;6:e18161. ArticlePubMedPMC
- 21. Fraser JG, Flood LM. An audiometric test for perilymph fistula. J Laryngol Otol 1982;96:513–20.ArticlePubMed
- 22. Hazell JW, Fraser JG, Robinson PJ. Positional audiometry in the diagnosis of perilymphatic fistula. Am J Otol 1992;13:263–9.PubMed
Citations
Citations to this article as recorded by

- Light cupula phenomenon: a systematic review
Nilüfer Bal, Melike Altun, Elif Kuru, Meliha Basoz Behmen, Ozge Gedik Toker
The Egyptian Journal of Otolaryngology.2022;[Epub] CrossRef









 KBS
KBS
 PubReader
PubReader ePub Link
ePub Link Cite
Cite